By T.J. Coles
This is part 11 in our series. For parts one to 10, click here.

Photo: APO
The latest document dump is arguably the most crucial, and thus the most censored. It should provide the raw data on the concentrations of antibodies in human blood stimulated by the injectable product.
But it doesn’t.
Readers will be alarmed to see hundreds and hundreds of blank pages, redacted mainly under section (b)(4) of the US Freedom of Information Act: the protection of trade secrets.
It would seem that Pfizer is hiding under (b)(4) to conceal the results of the injection trials, as well as its laboratory safety protocols.
If we had access to these data, we could interpret how well or poorly the alleged vaccine performed, and compare its performance to natural immunity.
THE BACKGROUND
In April, Pfizer released over 50 documents. Most of them come from the Pfizer Vaccine Research and Development laboratory in Pearl River, New York.
They concern the particulars of how the company analysed the results of its vaccine candidate trials.
But almost every single page is redacted, which renders rather meaningless the US judge’s order: that the documents enter the public domain.
So, what is the company hiding?
In August last year, Annaliesa Anderson became head of Pfizer’s Vaccine Research and Development.
Her previous work included leading the team of scientists that delivered the antiviral oral medication, Paxlovid, to emergency use authorization.
No sooner had Dr. Anderson taken up her new position, Nature published a shocking, non-clinical piece of public relations for Pfizer, authored by Dr. Anderson (details below).
The article was published just one month after Pfizer announced its plan to invest $470 million in its Pearl River vaccine facility; one of the many sites overseen by Anderson.
PEARL RIVER
April’s documents are dated from mid-2020 and concern analyses conducted by the company at one of the sites now overseen by Dr. Anderson, Pearl River.

Pearl River is located in Rockland County, New York, about half way between Philadelphia and Hartford, Connecticut, on the east coast.
Pfizer says that “Pearl River scientists are currently working to develop vaccines against other major infectious diseases.”
IgG is Immunoglobulin G, the most common human antibody. Assays are assessments.
Pfizer measured post-injection human antibodies by comparing them to the number of receptor-binding domain (RBD) proteins in the virus. RBDs form part of SARS-CoV-2’s spike that enable the virus to lock onto cells and infect them.
Pfizer assessed them using ThermoFisher Scientific’s Luminex machine.
WHAT’S MISSING?
The July 2020 document contains a Glossary which includes definitions, like Ag (antigen), LNB (laboratory notebook), and so on. But the definition of “Batch” is completely redacted.

A specialist site describes batch-testing as the method of conducting multiple tests in order to save money:
“One of the main advantages of batch testing is that it reduces costs.”
It also notes that such testing methods can “deliver more reliable generic results than if a sample is tested individually.”
In other words, if “Batch” refers to this method in the documents, then Pfizer likely cut corners by batch-testing and reported on generic instead of patient-specific results.
The definition of “dilution plating”–a technique used to estimate the number of specific organisms in a sample–is also redacted.
Is this also because Pfizer cut corners, risking potential sample contamination?
In one instance, an entire acronym and its definition are redacted.
REDACTED SAFETY PROCEDURES
Being a giant corporation partly responsible for people’s health, one might assume that Pfizer would be transparent about its safety protocols.
However, a link to the company’s Safety Data Sheets, which employees are meant to consult, is redacted. Why?
More strikingly, a link to the Pearl River Environmental Health and Safety Laboratory Manual is redacted.

Specific equipment names are also redacted.

Why redact those but not redact the Luminex product? Is it because the equipment used was below the optimum quality grade?
These include washing stations, plate washers, platform shakers, and equipment unknown because it has also been entirely redacted.
Buffers maximise the adsorption (adhesion of particles) of antigens onto plates for analysis. The entire section “Buffers” is redacted, as is half of the section on Procedure.
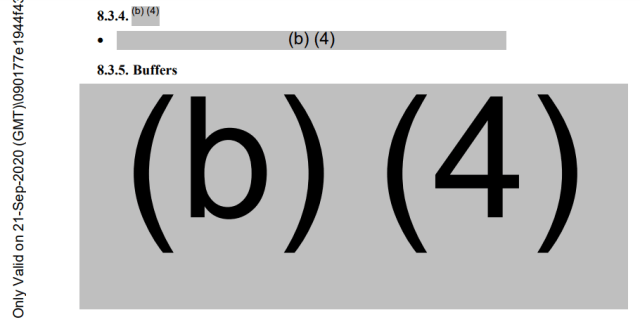

Microspheres are used for assaying. Presumably to protect patents, Pfizer redacted the entire section on microspheres.

The sections on robotic and manual preparations are redacted, as are the Reference Standards.



QSC is quality control serum. Perhaps most disturbing of all, a section entitled Unknown Specimen and QCS is entirely redacted.

Were the assessed samples contaminated? If so, with what and did this affect the outcomes of the analyses?
In a final insult, two names of the five signatories are redacted under section (b)(6): the right to privacy.

What right do public servants have to professional privacy when they are developing a potentially dangerous health product used by billions of people?
LIGHTSPEED, WARP SPEED,
THE SPEED OF SCIENCE
In the pro-pharma, Nature PR article mentioned above, Dr. Anderson acknowledges that pre-pandemic vaccine development and marketing used to take between 10 and 15 years.
In 2020, Paul Dabbar, Under Secretary of State at the US Department of Energy, said that American companies would work at the “speed of science” to develop a vaccine.
Thomas Gatliff, of Gatliff Technologies, quipped: The speed of science is:
“The velocity at which money flows from government bank accounts into the accounts of pharmaceutical company accounts.”
Dabbar’s language coincided with the US government’s effort to develop a vaccine, entitled Operation Warp Speed.
In the same vein, Dr. Anderson more recently described Pfizer’s approach as “lightspeed.”
Anderson says that Pfizer could get the injectable product out so quickly because government and Pfizer cleared away unnecessary bureaucracies, obliged different teams to work simultaneously instead of linearly, and based research and technology on decades-worth of prior research.
This new approach, she says, will enable new injectable mRNA products to get into human bodies quicker than ever before:
“[It] provides real-world lessons on how to help avoid a regression to pre-pandemic ways.”
SACRIFICING SAFETY
But “pre-pandemic” ways meant safety first via exercising the precautionary principle: hence the pulling from market of so many prior, failed vaccines.
In her article, Anderson leaves out a few things:
The Pfizer papers show that the company mixed up stages with phases, unblinded their trials, failed to follow up on participant health and mortality, statistically rounded injuries (in some cases to zero), and secretly planned for a third dose while telling the world that two shots were “safe and effective”.
If this is the method by which the company will move at “lightspeed” to bring new products to market, it might be better to go back to the previous method.
Pfizer says: “We strive to set the standard for quality, safety and value in the discovery, development and manufacture of health care products.”
But the latest batch of censored Pfizer papers does not allow us to verify this claim.
CONCLUSION
This article mainly quotes from the July 2020 document because, of all the Pearl River papers, it is one of the least redacted.
Many of the documents (particularly from February 2021) consist almost entirely of redacted pages.
I contacted Dr. Anderson to ask why she thought that the documents were so heavily censored.
She and colleagues at Pfizer did not reply in time to comment for this article.

Dr. T.J. Coles is the author of several books, including — The War on You.
Check out T.J’s Amazon page by clicking here.
View more TOTT content from Coles here.
KEEP UP-TO-DATE
For more TOTT News:
Facebook — Facebook.com/TOTTNews
YouTube — YouTube.com/TOTTNews
Instagram — Instagram.com/TOTTNews
Twitter — Twitter.com/EthanTOTT
Bitchute — Bitchute.com/TOTTNews
Gab — Gab.com/TOTTNews



Delivered “at the speed of science” by arguably one of the most evil organisations ever to exist on the planet. I present to you, ladies and gentlemen, THE QUAX.
Exactly, avoid anything that has the Pfizer name on it.
Aaron Siri Esq will sort this out…he’s having many wins! Walenski just resigned as the unoathed are shed from the CDC…things starting to turn I hope.